Kirsten Zornado
Evan Mark
9/15/14
Flame Test Lab
Purpose: To observe and identify colors of metallic ions when placed into a fire.
Procedure: In this lab, we will carefully combine certain (known and unknown) chemical ions with methanol, then set them on fire and observe the color they turn into.
Pre Lab Questions:
Q: What color of light is the lowest in energy?
A: Red, and it goes down in rainbow order.
Q: What color of light is the highest in energy?
A: Violet, and it goes up in rainbow order.
Q: What color of light is the highest frequency?
A: Violet
Q: What color of light is the lowest frequency?
A: Red
Q: How are electrons “excited”?
A: They are promoted to a higher energy state when the are excited.
Q: What does it mean when the electrons are “excited”?
A: It means that the electron is in a higher-energy state (an electron in a normal energy state is in ground state). Leaving this excited state means that the electron emits a photon of light.
Q: In your own words, write a short explanation of how an electron absorbs energy and re-emits it as light and why different elements have different spectra.
A: When an electron absorbs energy (either by fire or some other method), it goes into an "excited" state. To get back to its normal ("ground") state, it emits a photon of light. Different elements emit different colors of photons because they pick up different amounts of energy before they become "excited".
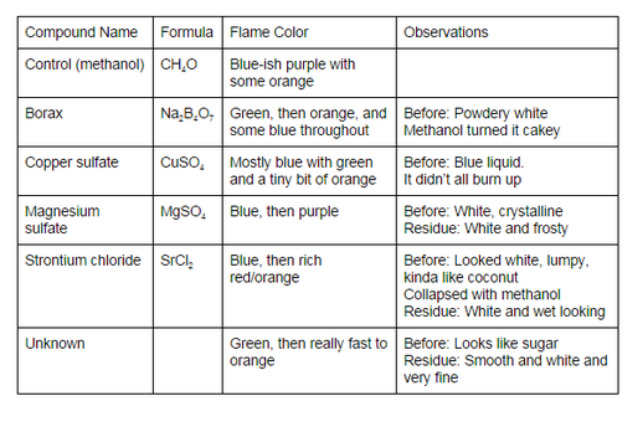
Data:
Evan Mark
9/15/14
Flame Test Lab
Purpose: To observe and identify colors of metallic ions when placed into a fire.
Procedure: In this lab, we will carefully combine certain (known and unknown) chemical ions with methanol, then set them on fire and observe the color they turn into.
Pre Lab Questions:
Q: What color of light is the lowest in energy?
A: Red, and it goes down in rainbow order.
Q: What color of light is the highest in energy?
A: Violet, and it goes up in rainbow order.
Q: What color of light is the highest frequency?
A: Violet
Q: What color of light is the lowest frequency?
A: Red
Q: How are electrons “excited”?
A: They are promoted to a higher energy state when the are excited.
Q: What does it mean when the electrons are “excited”?
A: It means that the electron is in a higher-energy state (an electron in a normal energy state is in ground state). Leaving this excited state means that the electron emits a photon of light.
Q: In your own words, write a short explanation of how an electron absorbs energy and re-emits it as light and why different elements have different spectra.
A: When an electron absorbs energy (either by fire or some other method), it goes into an "excited" state. To get back to its normal ("ground") state, it emits a photon of light. Different elements emit different colors of photons because they pick up different amounts of energy before they become "excited".
Data:
Conclusions:
In this lab, we learned about energy and light in electrons as they relate to fire. We also figured out that the unknown chemical was Borax, because of similarities between the two flame colors, and the way the raw chemical looked.
Post Lab Questions:
Q: Why do you think the chemicals have to be heated in the flame first before the colored light is emitted?
A: Because the fire energizes (excites) the electrons in the chemical.
Q: Most salts contain a metal and a non-metal. What type of bonds usually form between a metal and a non-metal?
A: Typically, Ionic Bonds form between metals and non-metals.
Q: What is the definition of a salt in chemistry?
A: A salt in chemistry is an ionic compound that is the result of the neutralization of an acid and a base.
Q: What is fire?
A: Fire is heat/light coming from a type of chemical reaction in which oxygen is combined with other elements, in this case, hydrogen and carbon, and a fuel, such as wood.
Q: What are the chemicals involved in lighting a match?
A: In safety matches (one with a matchbox that has a red "striking surface"), the match head has sulfur, powdered glass, and an oxidizing agent, often potassium chlorate, in it. The "striking surface" of the matchbox, where you strike the match to light it, has powdered glass, and red phosphorous on it. When you rub the match and "striking surface" together, the glass create friction, and a small spark. This spark sets the potassium chlorate up to break down, releasing oxygen. The sulfur is the fuel for the fire, along with the oxygen. The only difference between a safety match and a strike anywhere match is that, in the strike anywhere match, the red phosphorous is on the tip, so everything is all in one place.
Q: What is a chemical reaction?
A: A chemical reaction is when one substance reacts with another one, transforming into another substance.
Q: What natural cycles does wildfire have an impact on?
A: Wildfires have an affect on cycles such as the Carbon Cycle, and are affected by the El Nino, and El Nina cycles.
Q: List at least three impacts of wildfire in San Diego.
A: They clear out dead brush, kill plants and animals, and make everyone in the county (everyone near the fires, that is) freak out.