Ionic and Covalent Bonding
|
Ionic Bonding:
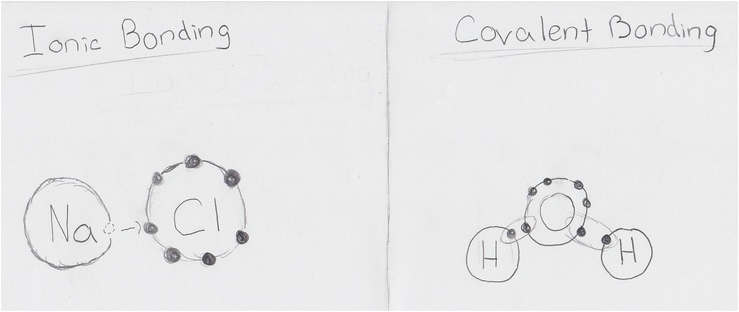
What types of atoms typically form ionic bonds? Typically, a metal and a nonmetal form ionic bonds How are ionic bonds formed and what type of structure do they create? They are formed when an electron transfers between two atoms with a large difference in electronegativity. Electronegativity is the atom’s ability to gain electrons. Typically, ionic bonds create ionic crystals. What are the typical properties of ionic substances? Include the following: physical state, melting point, solubility in water, electrical conductivity, and any other properties you’d like to include. Ionic substances typically have high melting and boiling points, conduct electricity, are a solid, and are soluble in water. An example of an ionic substance is NaCl, or salt. Its melting point is 1,474°F (801°C), it conducts electricity, it is solid, and is soluble in water. Create an image that you feel best represents ionic bonding. (See below) |
Covalent Bonding:
What types of atoms typically form covalent bonds? Typically, covalent bonds are formed by similar atoms, like metals to metals, and nonmetals to nonmetals. How are covalent bonds formed and what types of structure do they create? Covalent bonds are formed when two atoms with very similar electronegativity charges share electrons. They are typically stable molecules. What are the typical properties of covalent substances? Include the following: physical state, melting point, solubility in water, electrical conductivity and any other properties you’d like to include. Covalent substances can be gases, liquids, or solids. They tend to have very low melting and boiling points, and are poor electrical conductors. They are typically not soluable in water. Explain the difference between polar and nonpolar covalent bonds. Be sure to describe electronegative and the role it plays in determining which type of bond is formed. Polar covalent bonds are like nonpolar (regular) covalent bonds in that the atoms are sharing electrons, but the electrons are typically closer to one atom. This is caused by one of the atom’s electronegativity being slightly higher than the other’s. Not enough for that “stronger” atom to take it, but enough so that it has the electron closer. In nonpolar covalent bonds, the electronegativity is very close, neither of the atoms is “stronger”. Create an image that you feel best represents covalent bonding. (See below) |
Creative Product:
For my creative product, I made one panel comics based off of each of the bonding types.
For my creative product, I made one panel comics based off of each of the bonding types.