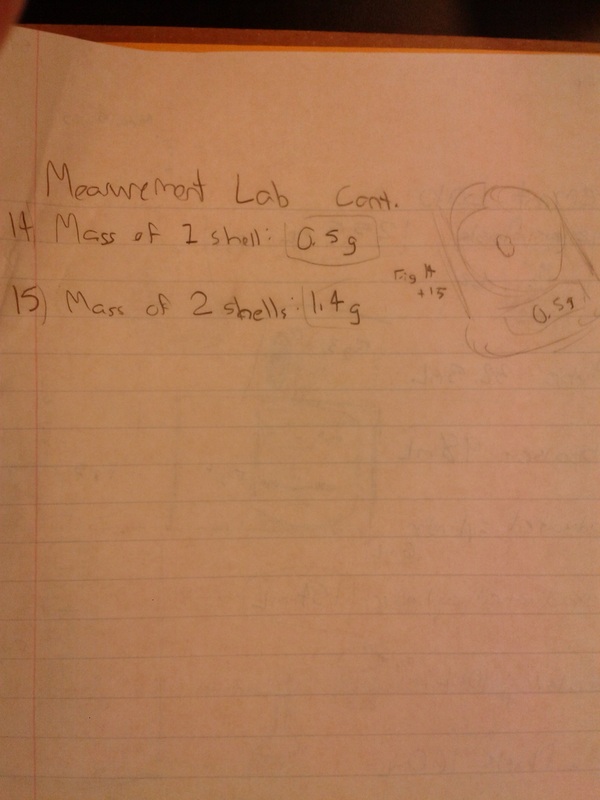On the first and second days of school, we did two labs. In one, we had to fit a gummy worm into a peach ring using only paper clips. In the other, we had to measure varying amounts of water inside of different objects.
Kirsten Zornado
Arjun Kraft
8/25/14
Save Fred
Purpose: The purpose of the lab is to save Fred (the gummy worm) from drowning.
Procedure: We had to use the paper clips given to us (four of them) to lift Fred into the peach ring (life preserver), without letting him "fall in the water" until he had in life preserver on.
Pre-Lab Questions:
Q: Why is it important in science to follow an agreed-upon procedure?
A: It is important because then all of the controllable things that are not being tested are consistent.
Q: Why is it important to document your procedure and any errors or differences?
A: It is important to document everything so that if something you did is off, you can correct it later, and know why what you did wrong was wrong.
Q: Why do we not normally have food in chemistry class?
A: Because many chemicals used in the class are poisonous, and if they are consumed, will make the consumer very sick.
Data/Calculations:
Fred's Mass: 7.6 g
Length: 8.2 cm
Life Preserver's Mass: 7.4 g = 74 mg
Diameter: 3.5 cm = 35 mm
Boat's Volume: 266 ml
Circumference: dπ = 8.4π = 26 cm
Conclusion: We saved Fred!
Post-Lab Questions:
Q: Was one of you more a leader, and the other followed, or did you share responsibility for ideas equally? Why did it work out that way?
A: I think I took more of a leading role, because my partner didn't really get started on working until I started suggesting what we could do.
Q: What areas of expertise can you bring to a project? (What are you good at?)
A: I'm not sure if I have any specific talents in chemistry, but if I do, I'm sure they will shine soon.
Q: What do you feel like you might struggle with in chemistry?
A: I might struggle with working with dangerous chemicals, because I get nervous around deadly things.
Arjun Kraft
8/25/14
Save Fred
Purpose: The purpose of the lab is to save Fred (the gummy worm) from drowning.
Procedure: We had to use the paper clips given to us (four of them) to lift Fred into the peach ring (life preserver), without letting him "fall in the water" until he had in life preserver on.
Pre-Lab Questions:
Q: Why is it important in science to follow an agreed-upon procedure?
A: It is important because then all of the controllable things that are not being tested are consistent.
Q: Why is it important to document your procedure and any errors or differences?
A: It is important to document everything so that if something you did is off, you can correct it later, and know why what you did wrong was wrong.
Q: Why do we not normally have food in chemistry class?
A: Because many chemicals used in the class are poisonous, and if they are consumed, will make the consumer very sick.
Data/Calculations:
Fred's Mass: 7.6 g
Length: 8.2 cm
Life Preserver's Mass: 7.4 g = 74 mg
Diameter: 3.5 cm = 35 mm
Boat's Volume: 266 ml
Circumference: dπ = 8.4π = 26 cm
Conclusion: We saved Fred!
Post-Lab Questions:
Q: Was one of you more a leader, and the other followed, or did you share responsibility for ideas equally? Why did it work out that way?
A: I think I took more of a leading role, because my partner didn't really get started on working until I started suggesting what we could do.
Q: What areas of expertise can you bring to a project? (What are you good at?)
A: I'm not sure if I have any specific talents in chemistry, but if I do, I'm sure they will shine soon.
Q: What do you feel like you might struggle with in chemistry?
A: I might struggle with working with dangerous chemicals, because I get nervous around deadly things.
Kirsten Zornado
8/26/14
Measurement Lab
Purpose: The purpose of this lab was to learn how to measure certain things in chemistry.
Procedure: We went to each station with our lab notebooks, and measured the item there.
Data:
8/26/14
Measurement Lab
Purpose: The purpose of this lab was to learn how to measure certain things in chemistry.
Procedure: We went to each station with our lab notebooks, and measured the item there.
Data:
- Erlenmeyer Flask: 260 ml
- Bottle: 260 ml
- Conical Tube: 32.5 ml
- 400 ml Beaker: 98 ml
- 10 ml Graduated Cylinder: 6ml
- 250 ml graduated cylinder: 154 ml
- 10 ml Beaker: 8 ml
- Volumetric Flask: 160 ml
- Erlenmeyer Flask: 1235 ml
- 600 ml Beaker: 22 degrees C
- 400 ml Beaker: 71 degrees F
- Length of a tray (long ways): 0.41 m
- Length between two blue marks: 4.4 cm
- Mass of one shell: 0.5 g
- Mass of two shells: 1.4 g
Conclusion: I learned how to measure things in all of the provided containers.